Cryptosporidiosis in Human and Animal a Systematic Review and Meta-analysis
- Review
- Open Admission
- Published:
Cryptosporidium fauna species in Islamic republic of iran: a systematic review and meta-analysis
Tropical Medicine and Health volume 48, Article number:97 (2020) Cite this article
Abstruse
Background
Cryptosporidiosis is an acute and short-term infection which can atomic number 82 to severe diarrhea (intestinal cryptosporidiosis) associated with a persistent cough in the host with immune system defect. This systematic review and meta-analysis was conducted to estimate the prevalence of animal Cryptosporidium species and the corresponding epidemiological aspects in Iran.
Methods
In this study, all original research articles relating to the animal cryptosporidiosis in Islamic republic of iran were collected from reliable databases using keywords. A meta-analysis was conducted separately for each subgroup, and heterogeneity among the studies was performed using the Q and I ii tests. Furthermore, it should be noticed that the significance level in the statistical analysis with the Comprehensive Meta-analysis software was considered to be less than 0.05. Finally, meta-analysis results were shown in forest plot with a 95% CI.
Results
In full, 4795 studies were included in the initial screening. Duplicated or not-original studies and the ones which did not meet our considered criteria were excluded from the list. Out of the 100 articles included in our get-go listing for the meta-assay, xl, 16, 13, 10, ix, 7, and five were washed on cattle and calves, birds, dogs, sheep, rodents, camels, and horses, respectively. The prevalence rate of cryptosporidiosis among the birds, horses, rodents, camels, dogs, cattle, and sheep in Islamic republic of iran was estimated to exist 7.5%, xix.5%, twenty.8%, 8.4%, iv.9%, 14.4%, and 9.1%, respectively.
Determination
The unlike Cryptosporidium species take been found in different regions of Iran. Geographical region, climate, and domestic animals are considered every bit factors responsible for animal cryptosporidiosis prevalence in the area. Moreover, this parasite is zoonotic which causes illness in animals besides as humans which tin can effect in economic loss.
Introduction
Abdominal parasites are considered an of import public health problem in humans and animals in developing and low-income countries [1,ii,3]. Cryptosporidium is i of the most mutual abdominal protozoan parasites which is located in the phylum of Apicomplexa and causes cryptosporidiosis [2]. Cryptosporidium is spread easily in the surround due to its unproblematic transmission via contaminated h2o, air, and dust. Cryptosporidiosis is considered as a major economic problem in many countries including Iran, and at that place are annually numerous reports of this infection in immunocompromised and young children [two,3,iv,5]. Different methods have been used to detect the protozoan parasite including the molecular diagnostic method which is considered ane of the about useful diagnostic tools.
This method has identified up to 30 species and more 50 genotypes of Cryptosporidium [6,7,8]. Dissimilar species of Cryptosporidium accept been reported in diverse hosts including birds, horses, cattle, sheep, camels, rodents, and dogs. Cryptosporidium parvum, C. hominis, C. canis, C. felis, C. meleagridis, and C. muris were distinguished from gastrointestinal diseases likewise every bit diarrhea in humans [9]. Notwithstanding, the infection typically occurs in a brusque-term and acute form in immunocompromised and HIV-positive individuals. In these cases, it tends to remain in the lower intestine for up to half dozen weeks with astringent diarrhea and persistent cough [9]. This protozoan could develop its life cycle in one host without the requirement of other animals as intermediate or reservoir hosts [9, 10]. Despite the many studies which were conducted in Iran on investigating the prevalence of cryptosporidiosis in different animal hosts, these data accept non shown the overall prevalence of animals in Iran. Since cryptosporidiosis causes irreversible economic damages to domestic animals, critical screening programs and epidemiological aspects should be considered by authorities. This systematic review and meta-analysis was conducted to gauge the prevalence of animal Cryptosporidium species and their epidemiological aspects in Iran.
Materials and methods
Study protocol
The present systematic review focused on the estimates of the prevalence of animate being Cryptosporidium species according to the PRISMA guidelines for systematic review and meta-analysis [11] (Supplementary one).
Search strategy
In order to select the suitable articles for this study, all records since 1991 up to February 2018 were investigated using seven international databases in English including PubMed, Web of Science, Scopus, Science Direct, and Google Scholar search engine as well as national databases in Persian including Magiran (http://www.magiran.com/) and Scientific Information Database (SID) (http://world wide web.sid.ir/). Furthermore, references of each article were screened manually, and the authors were contacted for additional references.
The databases screening was performed using the post-obit keywords: prevalence, Cryptosporidium, cryptosporidiosis, animate being Cryptosporidium, Cryptosporidium species, C. parvum, C.hominis, animal, cattle, dogie, sheep, caprine animal, camel, horse, rodent, bird, craven, epidemiology, Iran, serology, PCR, and molecular (Box 1).
Eligibility criteria
All original descriptive studies which investigated the prevalence of Cryptosporidium in animals in Iran, both in English language and Persian, were included in this study. Duplicates, qualitative studies, review manufactures, case reports, case series, and studies out of Iran or those performed on humans were excluded. Finally, manufactures with epidemiological parameters of interest were selected, and a total of 100 nerveless articles fulfilled the considered criteria.
Quality assessment
The scoring organisation based on the 8-detail modified Newcastle Ottawa Scale (NOS) for non-randomized studies was used for assessing the quality of the studies. In this system, each question has a score betwixt 0 and i, and the maximum signal summation is 9. Studies with signal summation 5 or less, half dozen–7, and 8–9 were considered depression, moderate, and loftier quality, respectively [12].
Screening and information extraction
All records were evaluated based on their title and abstract and according to the inclusion and exclusion criteria by ii researchers (MM and NB) independently. The kappa index showed an agreement of 91% between the findings of ii researchers. The total-text version of the papers was obtained through library resource and online databases. Finally, the departure between records amidst the researchers was corrected past re-examining the manufactures. The understanding was reached by group discussion with a 3rd researcher (SK).
Data extraction was conducted independently by 2 researchers (MM and NB) and imported to the pre-prepared class. Data including authors, year of study, publication year, kind of creature, geographical surface area of the study, number of examined, number of positive, prevalence charge per unit, and type of host were extracted from articles.
Quality assessment studies
The methodological quality of the studies was examined based on the guidelines of the Newcastle and Ottawa Statements [12]. This guideline sets the criteria for selecting people to study, comparison and accepting them, likewise every bit exposure and consequences where a maximum of nine stars tin be allocated to each study. Studies with 7 stars or more are classified as high-quality studies, and studies with 6 stars and less are considered equally depression-quality studies. Investigating the probability of an error in the results of the studies is performed separately by two researchers. The disagreement betwixt the parties is resolved through negotiations.
Data analysis
The meta-assay method was adopted to a 95% confidence interval (CI) in order to assess the pooled prevalence of Cryptosporidium infection in animals using the random effect model. Various subgroup analyses were separately conducted based on animal type and the associated species. Finally, meta-analysis results were displayed in forest plot (reported as consequence estimates (ES) with a 95% CI). We besides performed a sensitivity assay to verify the stability of the data. In lodge to assess the sensitivity analysis, the outcome judge was estimated irrespective of one study at a time, and the robustness of the pooled estimate was assessed. Heterogeneity was calculated among the studies by the Q and I two tests [6, viii]. Cochran'due south Q test (Q statistic, p < 0.10) showed statistically pregnant heterogeneity, and I 2 statistic (I two > fifty%) indicates a large heterogeneity. Statistical analysis and information analysis were performed using the 2d version of the Comprehensive Meta-assay software. The significance level was considered to be less than 0.05.
Results
Description of studies
In total, 4795 studies were collected in the initial screenings from the published articles and their references in the screened databases up to February 2018. Specifically, 62, 124, 174, 62, 543, 10, and 3820 studies nerveless from PubMed, Web of Science, Scopus, Scientific Data Database, Magiran, Science Direct, and Google Scholar, respectively. A full of 1933 duplicated records and v studies which were not original articles (i.eastward., letter, commentary, review) were screened out. Regarding the relevance of the title and abstruse to the purpose of the written report, 2857 irrelevant studies were excluded. Accordingly, 2757 studies were retrieved for further assessment. Altogether, 100 articles were selected for the meta-analysis study. The display procedure and literature search results were presented respectively in Fig. 1 and Tabular array 1.
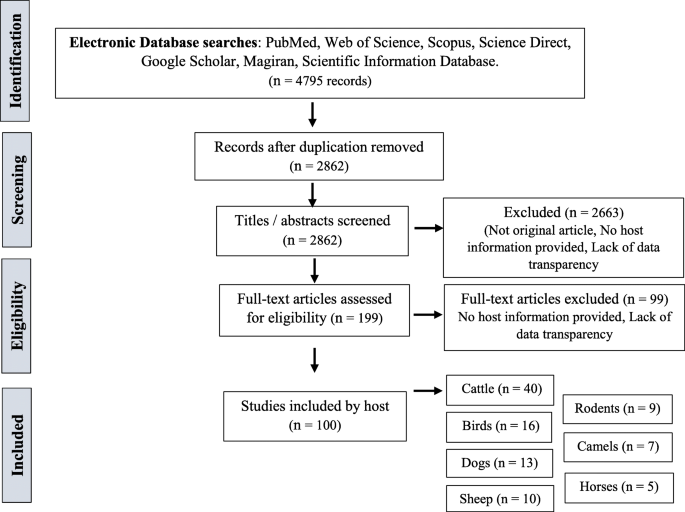
Flowchart describing the study design process
Within these 100 articles, xl, xvi, 13, x, 9, vii, and five studies were performed on cattle, birds, dogs, sheep, rodents, camels, and horses, respectively. The most frequent studies were performed on cattle and the least ones on horses. Because the various projects in searching cryptosporidiosis on cattle in Iran, the distribution of positive cases relating to cattle is presented in Fig. 9. The quality assessment of studies using the guideline of the Newcastle Ottawa Scale showed that 27%, 65%, and 8% of the studies have low, medium, and high quality, respectively.
Main analysis
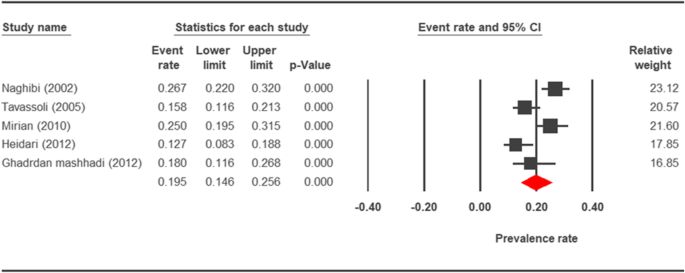
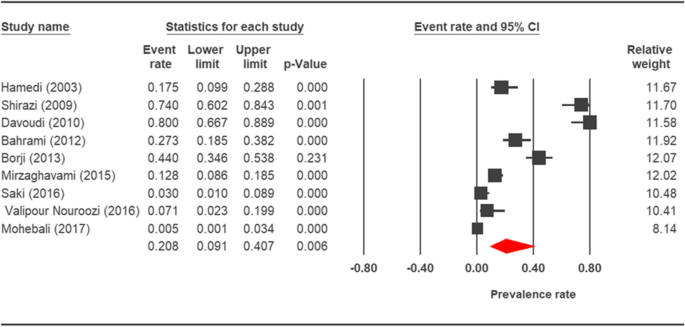
The prevalence charge per unit of cryptosporidiosis within a 27-year menses for birds, horses, rodents, camels, dogs, cattle, and sheep in Iran using the random issue model was estimated to be 7.5% (95%, CI = 4.7%, 11.9%), 19.five% (95%, CI = fourteen.6%, 25.half-dozen%), twenty.8% (95%, CI = 9.1–40.vii%), eight.4% (95%, CI = 3.8%, 17.8%), 4.ix% (95%, CI = two.6%, 8.8%), xiv.4% (95%, CI = eleven%, 18.6%), and 9.one% (95%, CI = viii.4%, 9.ix%), respectively. The forest plot diagrams of the current study are shown in Figs. 2, 3, four, 5, 6, 7, and 8.
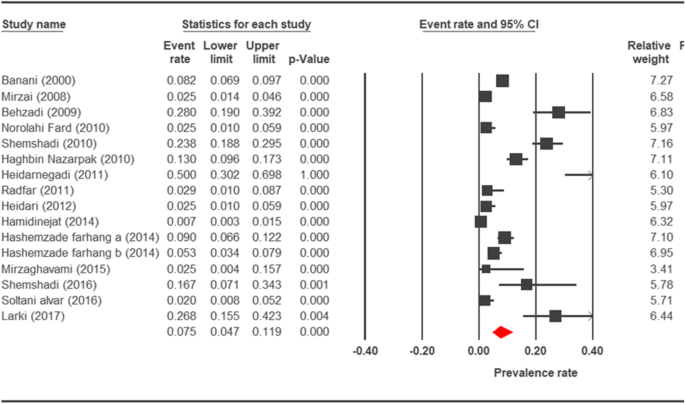
Forest plot diagram showing the prevalence charge per unit of Cryptosporidium infection in birds of Iran
Forest plot diagram showing the prevalence rate of Cryptosporidium infection in horses of Islamic republic of iran
Forest plot diagram showing the prevalence rate of Cryptosporidium infection in rodents of Iran
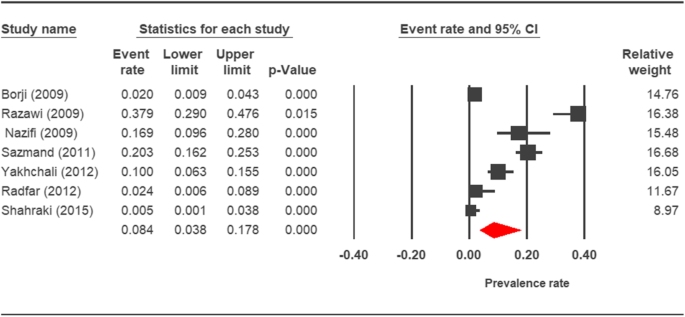
Forest plot diagram showing the prevalence rate of Cryptosporidium infection in camels of Iran
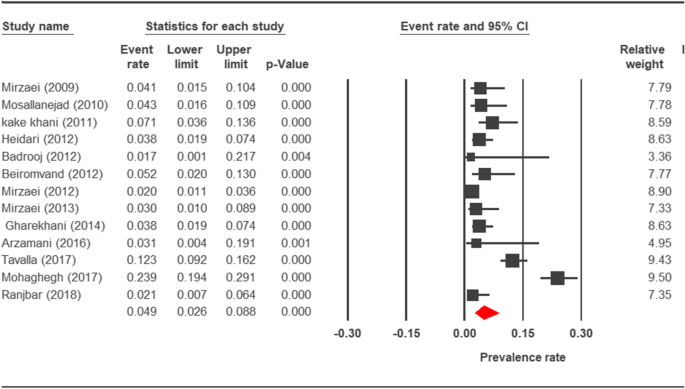
Wood plot diagram showing the prevalence rate of Cryptosporidium infection in dogs of Iran
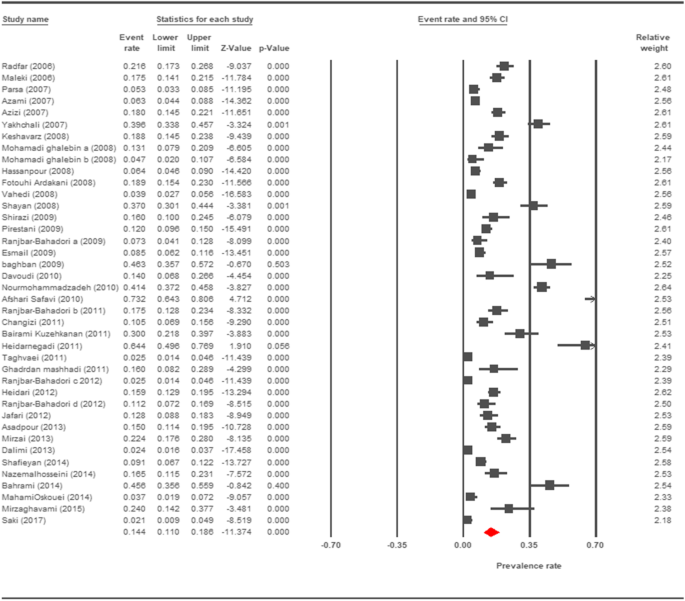
Forest plot diagram showing the prevalence charge per unit of Cryptosporidium infection in cattle of Iran
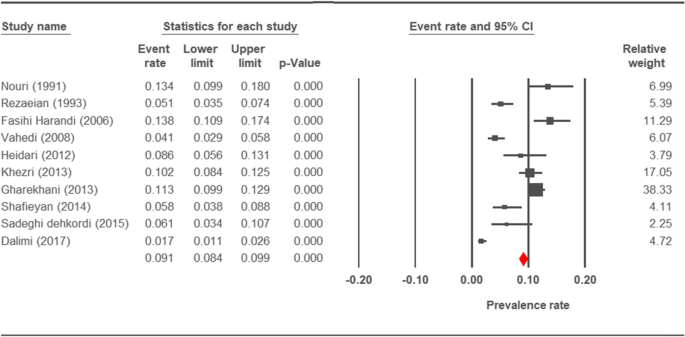
Forest plot diagram showing the prevalence rate of Cryptosporidium infection in sheep of Iran
A wide variation was observed in the prevalence estimations among the various studies. The Q statistic, df, and I 2 were as follows: 26.63, 15, and 43.74% for birds; xv.15, viii, and 47.18% for rodents; 11.15, half-dozen, and 46.17% for camels; 58.85, 43, and 26.93% for cattle; and xvi.64, 10, and 39.93% for sheep, respectively. Depression heterogeneity was reported in studies which were conducted on horses as well equally the ones on dogs. The statistic factors (Q statistic, df, and I two) were 3.86, 4, 0.00% for horses and 6.07, 12, and 0.00% for dogs.
The prevalence rate of Cryptosporidium infection in cattle is shown in Fig. nine. The most positive cases of cryptosporidiosis were reported in cattle of Due west Azerbaijan, Tehran, Khuzestan, Chaharmahal and Bakhtiari, Kohgiluyeh and Boyer-Ahmad, and Kerman provinces. Considerable positivity rates of cryptosporidiosis in cattle were identified in Razavi Khorasan, S Khorasan, Semnan, Hamadan, Alborz, and Eastward Azerbaijan provinces. There were no positive reports of cryptosporidiosis in cattle in other provinces.
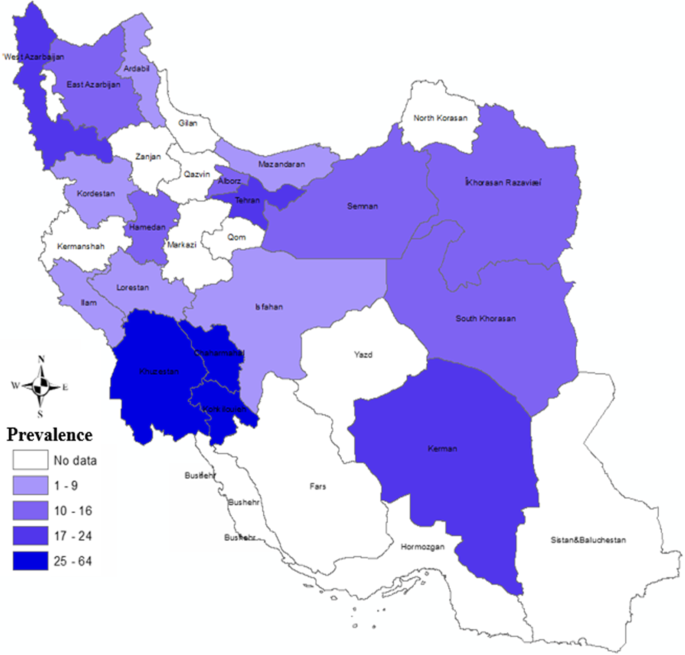
Incidence of Cryptosporidium infection in the Iranian cattle in different provinces [2]
Discussion
Cryptosporidiosis is one of the most important zoonotic diseases which is reported in humans and animals with a worldwide distribution in more than 106 countries and especially in developing countries [91, 92]. To the best of our cognition, this is the starting time systematic review and meta-analyses on the prevalence of beast cryptosporidiosis in Iran.
The nowadays study showed that the average prevalence rate of cryptosporidiosis in birds was 7.v% in Iran. Additionally, the prevalence rate of cryptosporidiosis in animals in Ahvaz, southwestern of Islamic republic of iran was reported as 50% [86] while it was shown that in Gilan, n of Iran, this charge per unit was 17% [84]. In a report by Jasim and Marhoon, it has shown that in Iraq, which is a neighboring region of Iran, the cryptosporidiosis prevalence rate in wild and domestic birds was 58.1% [93]. Fifty-fifty though this region is about the southwest of Iran, the prevalence was higher than in Iran. Moreover, a prevalence of 49% was shown in Mexico [94], and in Brazil, 76% of birds were infected past Cryptosporidium [95]. Changes in the prevalence seen in various reports indicate that the probability of transmitting the parasite is higher among animals living together on farms and next to each other compared to other studies that have examined private specimens.
Preventive efforts by Iranian government related to awareness of zoonotic diseases, control of stray dogs, and a depression population of pet dogs have increased the possibility of transmission of the disease from livestock [96]. On the one mitt, the stray dogs are the largest group of dogs in both rural and urban areas in Iran which normally become infected by roaming in homo neighborhoods and feeding on contaminated residues. On the other paw, domestic dogs are not restricted to the express expanse of houses or farms. Stray dogs are allowed to wander around, so it increases the adventure of zoonotic infections in rural habitats. In this study, the overall prevalence of cryptosporidiosis between dogs was found to be 4.9% in Iran. There are various reports of cryptosporidiosis prevalence in the different geographical regions of Iran. Mohaghegh et al. reported a prevalence of 21.7% and 25.four% of cryptosporidiosis respectively in domestic and stray dogs of Kermanshah [61]. Furthermore, the 12.3% prevalence rate of cryptosporidiosis in dogs was observed in Ahvaz [threescore]. These results are higher than the information which were obtained in other regions of Islamic republic of iran, specifically v% in Chenaran, northeast of Islamic republic of iran [56]; seven% in Ilam [97]; 2% in Kerman, southeast of Iran [98]; 2.ix% in Urmia, northwest of Islamic republic of iran [99]; two.14% in Isfahan, centre of Iran [62]; and three.8% in Hamadan, west of Iran [17]. The high prevalence of cryptosporidiosis in some areas, for instance, Kermanshah Province, indicated that humans are at serious risk of Cryptosporidium infection. Furthermore, the infection tin spread vastly and cause severe bug in the customs.
Epidemiological studies on cryptosporidiosis infection indicated that the prevalence of Cryptosporidium species in dogs is very different in various countries changing from 0 to 52.seven%. These differences might be attributed to several factors, such as geographical area, sample size, keeping a domestic dog, correlation with other hosts (such equally goat, sheep, horse, cattle, and pig), dissimilar species of Cryptosporidium, and sampling procedures also as diagnostic methods [100, 101]. The current results imply that the prevalence rate of cryptosporidiosis in Iran is higher than in countries such as the Czech republic with 1.4% [102], Thailand with two.i% [103], Brazil with 2.four% [104], Japan with three.9% [105], and Spain with 4.1% [106], just lower than in Nigeria with xviii.five% [107] and Romania with 52.vii% [108].
Rodents could exist potential reservoir hosts for zoonotic cryptosporidiosis. During extensive epidemiological studies that accept been performed throughout the world, infection in rodents was highly varied from 7.half-dozen% in Maryland [109] to 63% in Britain [110]. Other studies showed unlike statistic in different countries. Specifically, 8.2% in northern Australia [111], eleven/five% in People's republic of china [112], 24.3% in Italy [113], 25.8% in Philippines [114], and 32.8% in the Usa of America [115] were reported. The average prevalence rate of cryptosporidiosis in rodents in Iran was estimated every bit twenty.8% in this study. Similar studies in different geographical regions of Iran showed diverse range of prevalence. The frequency of rodent's cryptosporidiosis in Meshgin shahr, Tehran, Shooshtar, and Ahvaz was 0.5% [75], 27.3 % [71], vii.1% [74], and 3% [1], respectively. In the parasite investigation of rodents of Mashhad, none resulted to be were contaminated (0%) [72].
The average prevalence of cryptosporidiosis in sheep was establish as nine.1%. Prevalence of cryptosporidiosis was reported every bit ane.69%, 5.viii%. six.ane% and viii.6% in Tehran [xx], Lorestan [18], Sanandaj [19] and Hamadan [xv], respectively. Majewska et al constitute like results in the west-central region of Poland (10.1%) [116] but lower rates were detected in Australia (24.5%) [117], and China (4.8%) [118].
The prevalence rate of cryptosporidiosis in cattle was 1.5% in Japan [119], 35.7% in Vietnam [120], 20.6% in Turkey [121], 40.6% in Canada [122], and 40.half-dozen% in the USA [123].
Still, according to this systematic review and meta-analysis, the prevalence of cryptosporidiosis in cattle and calves was 14.4% in Iran, and the prevalence rate in various geographical regions was equally follows: two.1% in Ahvaz [73], ix.07% in Lorestan [18], 16.45% in Qazvin [26], 22.iii% in the city of Urmia [48], and 28.iii% in the city of Mashhad [47]. Furthermore, our written report showed that the prevalence rate of cryptosporidiosis in camels and horses was 8.4% and 19.5%, respectively.
It was suggested in this study that the distribution of Cryptosporidium differs among geographical regions. Therefore, the study location might be one of the nearly determinant factors in cryptosporidiosis distribution. The highest prevalence rate (50%) of cryptosporidiosis was observed in Khuzestan Province [86]. This high prevalence might be attributed to the high temperature and humidity of the southwestern regions of Islamic republic of iran as well every bit the people's lifestyle, who have a high level of seafood consumption compared to other regions. Additionally, the immigration of birds to the south of Khuzestan Province may transmit parasite protozoan infection.
Conclusion
The relatively high prevalence of cryptosporidiosis infection among animals in Islamic republic of iran, more often than not among sheep, cattle, and calves, shows the enzootic status of cryptosporidiosis in the investigated areas and may be a threat to the inhabitants. Our data offer important information about the epidemiology of cryptosporidiosis among animals in Iran, which could be useful for managing and decision-making programs for the illness. Further investigation and monitoring will be required to expand the surveillance and control policies in order to reduce the prevalence of Cryptosporidiosis among livestock and consequently decrease the economical damages and public health hazards in Iran.
Availability of information and materials
Input data for the analyses are available from the respective author on asking.
Abbreviations
- HIV:
-
Human being immunodeficiency viruses
- CI:
-
Confidence interval
References
-
Saki J, Foroutan-Rad Thou, Asadpouri R. Molecular label of cryptosporidium spp. in wild rodents of southwestern Iran using 18 south rRNA gene nested-PCR-RFLP and sequencing techniques. J Trop Med. 2016;2016:1–six.
-
Mousavi-Hasanzadeh M, Sarmadian H, Ghasemikhah R, et al. Evaluation of toxoplasma gondii infection in western Islamic republic of iran: seroepidemiology and risk factors analysis. Trop Med Wellness. 2020;48:35.
-
Ghasemikhah R, Tabatabaiefar MA, Shariatzadeh SA, Shahbazi A, Hazratian T. A PCR-based molecular detection of strongyloides stercoralisin human stool samples from Tabriz city, Iran. Sci Pharm. 2017;85(two):17.
-
Mor SM, Tzipori S. Cryptosporidiosis in children in sub-Saharan Africa: a lingering challenge. Clin Infect Dis. 2008;47(7):915–21.
-
Tzipori S, Widmer 1000. A hundred-year retrospective on cryptosporidiosis. Trends Parasitol. 2008;24(4):184–nine.
-
Dalimi A, Tahvildar F. Molecular study on Cryptosporidium andersoni strains isolated from sheep based on 18S rRNA gene. Infect Epidemiol Microbiol. 2017;3(3):100–3.
-
Mirian S, Asadi 1000, Ferdowsi H, Rezakhani A. A survey on equus caballus cryptosporidial infection in Tehran Province. Arch Razi Inst. 2010;65(1):45–7.
-
Haghi MM, Etemadifar F, Fakhar Thou, Teshnizi SH, Soosaraei Chiliad, Shokri A, et al. Status of babesiosis among domestic herbivores in Iran: a systematic review and meta-assay. Parasitol Res. 2017;116(four):1101–9.
-
Ranjbar R, Mirhendi H, Izadi Thousand, Behrouz B, Mohammadi MR. Molecular identification of cryptosporidium spp. in Iranian dogs using seminested PCR: a first report. Vector Borne Zoonotic Dis. 2018;18(2):96–100.
-
Soosaraei M, Haghi MM, Etemadifar F, Fakhar M, Teshnizi SH, Hezarjaribi HZ, et al. Status of theileriosis amidst herbivores in Iran: a systematic review and meta-analysis. Vet World. 2018;xi(3):332.
-
Moher D, Altman DG, Liberati A, Tetzlaff J. PRISMA statement. Epidemiology. 2011;1(22):128.
-
Wells GA, Shea B, O'Connell D, Peterson J, Welch V, Losos M, et al. Championship of subordinate document. In: The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ontario: Ottawa Infirmary Enquiry Institute; 2009. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed 15 Jan 2009.
-
Nouri M, Karami M. Asymptomatic cryptosporidiosis in nomadic shepherds and their sheep. J Inf Secur. 1991;23(3):331–3.
-
Rezaeian M, Shahmoradi A, Dalimi A. Cryptosporidum in sheep as a source for human infection. Med J Islam Repub Iran. 1993;half-dozen(iv):273–4.
-
Fasihi Harandi 1000, Fotouhi AR. Cryptosporidium infection of sheep and goats in Kerman: epidemiology and hazard factor analysis. J Vet Res. 2008;63(i):47–51.
-
Vahedi N, Asl A, Saadat M. Primary research on gastro-intestinal cryptosporidium incidence charge per unit in lambs and calves in Amol city. Iran J Vet Res. 2009;64(2):101–3.
-
Heidari H, Gharakhani J. Study of cryptosporidium infection in the livestock (cattle, sheep, dogs, fowls) and humans, in Hamadan city and its suburbs during 2006-2011. Avicenna J Clinical Med. 2012;19(iii):67–74.
-
Khezri M, Khezri O. The prevalence of cryptosporidium spp. in lambs and goat kids in Kurdistan, Iran. Vet Earth. 2013;half dozen(12):974.
-
Gharekhani J, Heidari H, Youssefi M. Prevalence of cryptosporidium infection in sheep in Iran. Turkiye Parazitol Derg. 2014;38(1):22.
-
Shafieyan H, Alborzi A, Hamidinejat H, Tabandeh MR, Hajikolaei MRH. Prevalence of cryptosporidium spp. in ruminants of Lorestan Province, Islamic republic of iran. J Parasit Dis. 2016;xl(four):1165–nine.
-
Sadeghi Dehkordi Z, Partoandazan A, Abdolmaleki N. Epidemiology of cryptosporidiosis in sheep of Sanandaj city. Zoonoses Res. 2016;two(1):25–9.
-
Radfar 1000, Molaei K, Baghbannejad A. Prevalence of cryptosporidium spp. oocysts in dairy calves in Kerman, southeastern Iran. IJVR. 2006;7(2):81–4.
-
Maleki S, Nayebzadeh H. A survey on prevalence rate of cryptosporidiosis amidst diarrheic and healty cattle and calves in Khoram-Abad, Iran. J Vet Res. 2008;62(half dozen):423–6.
-
Parsa A. Prevalence of Cryptosporidium muris–similar (andersoni) infection in cattle and buffalo slaughtered in Tabriz surface area abattoir. Large Animal Clin Sci Res. 2007;1(ii):49–55 J. Vet. Sci.
-
Azami M. Prevalence of cryptosporidium infection in cattle in Isfahan, Iran. J Eukaryot Microbiol. 2007;54(1):100–ii.
-
Azizi Hamidreza PM, Batol D, Hosein R. Prevalence of Cryptosporidium parvum parasites in dairy calves less than one year sometime. Islamic republic of iran J Vet Med. 2008;3(4):96–8.
-
Yakhchali One thousand, Gholami E. Prevalence of Eimeria and cryptosporidium spp in cattle of Sanandaj urban center (Kurdistan province), Iran. J Pajouhesh Sazandegi. 2008;87:81–vii.
-
Keshavarz A, Haghighi A, Athari A, Kazemi B, Abadi A, Mojarad EN. Prevalence and molecular characterization of bovine cryptosporidium in Qazvin province, Islamic republic of iran. Vet Parasitol. 2009;160(3-four):316–viii.
-
Mohammadi Ghalehbin B, Arzanlou M, Fallah E, Kazemi A, Asgharzadeh M. Molecular identification of cryptosporidium sp. in the cattle stool samples in Ardabil city, northwestern Iran. J Anim Vet Adv. 2008;seven(three):246–50.
-
Hassanpour A. Prevalence of cryptosporidiosis in calves and humans to be in contact them in Tabriz expanse in Islamic republic of iran. Int J Infect Dis. 2008;12:e126.
-
Fotouhi Ardakani R, Fasihi Harandi G, Solayman Banai Southward, Kamyabi H, Atapour M, Sharifi I. Epidemiology of cryptosporidium infection of cattle in Kerman/Iran and molecular genotyping of some isolates. JKMU. 2015;xv(4):313–twenty.
-
Shayan P, Ebrahimzadeh E, Mokhber-Dezfouli M-R, Rahbari S. Recombinant Cryptosporidium parvum p23 as a target for the detection of cryptosporidium-specific antibody in calf sera. Parasitol Res. 2008;103(5):1207–11.
-
Davoudi J, Shirazi S, Mostofi S, Vahidi MS. Evaluation of cryptosporidiosis in human, calf and mice effectually Tabriz. Vet Microbiol. 2010;half dozen(1):75–9.
-
Pirestani M, Sadraei J, Dalimi AA. A survey on prevalence rate of cryptosporidial infection of farms in Shahriar County of Tehran and its aseptic importance in human. Vet Microbiol. 2010;85:44–53.
-
Ranjbar-Bahadori S, Sangsefidi H, Shemshadi B, Kashefinejad Thousand. Cryptosporidiosis and its potential risk factors in children and calves in Babol, north of Iran. Trop Biomed. 2011;28(i):125–31.
-
Fallah E, Ataallah H. Prevalence of cryptosporidium oocysts from calves in Kurdistan Province, of Iran. J Parasit Dis. 2009;4(2):1–5.
-
Baghban Farshad MM. Frequency of cryptosporidium oocysts in the midst of calves (cryptosporidium spp. oocysts) under ane month with diarrhea in Yasuj urban center. Vet J Islamic Azad University. 2010;6(iii):17–22.
-
Nourmohammadzadeh F, Davoodi Y, Jamali R, Nowrouzian I. Epidemiological written report on cryptosporidiosis in newborn calves in eastern Azarbaijan Province. J Vet Res. 2010;65(iii):247–70.
-
Safavi Afshari E, Mohammadi GR, Naghibi A, Rad M. Prevalence of cryptosporidium spp. infection in some dairy herds of Mashhad (Iran) and its association with diarrhea in newborn calves. Comp Clin Pathol. 2011;20(two):103–7.
-
Ranjbar-Bahadori S, Aliari M. Cryptosporidiosis and its adventure factor in calves of husbandries effectually of Tehran, Iran. In: Animal hygiene and sustainable livestock production. Vienna: Proceedings of the XVth International Congress of the International Lodge for Animal Hygiene; 2011. https://www.cabdirect.org/cabdirect/abstruse/20113296231.
-
Changizi E, Salimi-Bejestani One thousand, Vayeghan A. The cryptosporidium ryanae infection commence in Iranian cattle. J Vet Res. 2012;67(2):127–33.
-
Bairami Kuzehkanan A, Rezaeian M, Zeraati H, Mohebali Yard, Meamar AR, Babaei Z, et al. A sensitive and specific PCR based method for identification of cryptosporidium sp. using new primers from 18S ribosomal RNA. Iran J Parasitol. 2011;6(four):one–7.
-
Heidarnegadi S, Mohebali Yard, Maraghi S, Babaei Z, Farnia S, Bairami A, et al. Cryptosporidium spp. infection in man and domestic animals. Iran J Parasitol. 2012;7(1):53.
-
Ranjbar S, Azizzadeh M, Taghvaei M. Study on the infection rate to cryptosporidium in suckling calves of Ghuchan district. Iran J Vet Res. 2013;9(3):62–8.
-
Jafari MA, Ranjbar BS, Sheybani M. Investigating the possible role of protozoa (cryptosporidium, giardia coccidia) in diarrheal calves of Garmsar. Qom: Start National Congress of Dairy, Dairy and Related Industries; 2012. http://meditech.ir/conference.National.Congress.of.dairy.cattle. Accessed 5 Dec 2019.
-
Heydari Heydar GJ. Frequency of cryptosporidium infection in human, creature and poultry in Hamadan and suburbs over the years 2007-2012. J Res Health Sci. 2013;19(3):67–74.
-
Ranjbar-Bahadon S, Toni Due south. Infection to cryptosporidium in diarrheic calves: a provincial written report in southern Khorasan. J Vet Res. 2013;68(one):thirteen–9.
-
Jafari R, Maghsood AH, Fallah M. Prevalence of cryptosporidium infection among livestock and humans in contact with livestock in Hamadan district, Iran, 2012. J Res Wellness Sci. 2012;29(13):86–9.
-
Asadpour M, Razmi Thou, Mohhammadi K, Naghibi A. Prevalence and molecular identification of cryptosporidium spp. in pre-weaned dairy calves in Mashhad surface area, Khorasan Razavi Province, Islamic republic of iran. Iran J Parasitol. 2013;8(4):601.
-
Mirzai Y, Yakhchali G, Mardani K. Cryptosporidium parvum and Cryptosporidium andersoni infection in naturally infected cattle of Northwest Islamic republic of iran. Vet Res Forum. 2014;5(i):55–threescore.
-
Dalimi A, Tahvildar F, Kazemi B. Molecular identification of Cryptosporidium andersoni in Shahriar calves. Vet Enquiry Biol Pro. 2015;28(2):24–30.
-
Mojarad N, Nejad MR, Keshavarz A, Taghipour N, Lamuki RM, Salehi A, et al. Molecular characterization of bovine cryptosporidium using cryptosporidium oocyst wall protein (COWP) cistron. J Paramed Sci. 2014;5(3):17–20.
-
Bahrami S, Alborzi R, Molayan PH, Purbaram Southward, Mousavi B. Prevalence of cryptosporidium spp. infection and its association with diarrhea in buffalo calves in Khuzestan, a southwestern province of Islamic republic of iran. Buffalo Bull. 2014;33(4):393–9.
-
Oskouei MM, Fallah E, Ahmadi M, Safaiyan A, Bakhtiyari S, Naserifar R, et al. Molecular and parasitological study of cryptosporidium isolates from cattle in Ilam, west of Iran. Iran J Parasitol. 2014;nine(3):435.
-
Mirzaghavami M, Sadraei J, Forouzandeh Thousand. Detection of cryptosporidium spp. in gratuitous ranging animals of Tehran, Iran. J Parasit Dis. 2016;xl(4):1528–31.
-
Mosallanejad B, Hamidinejat H, Avizeh R, Ghorbanpoor Najafabadi M, Razi JM. Antigenic detection of Cryptosporidium parvum in urban and rural dogs in Ahvaz district, outhwestern Islamic republic of iran. Iran J Vet Res. 2010;xi(3):273–8.
-
Kakekhani S, Bahrami AM, Ahmady Asabchin S, Doosti A. A study on protozoan infections (giardia, Entamoeba, Isospora and cryptosporidium) in stray dogs in Ilam Province. JVCP. 2011;5(3):1325–30.
-
Badrooj A, Asgari K, Hatam Thousand. Molecular survey on cryptosporidiosis in stray dogs and cats in shiraz city during 2011-2012; 2012. http://elib.sums.air conditioning.ir/cgi-bin/koha/opac-main.pl. Accessed xx December 2019.
-
Beiromvand M, Akhlaghi 50, Massom SHF, Meamar AR, Motevalian A, Oormazdi H, et al. Prevalence of zoonotic intestinal parasites in domestic and stray dogs in a rural expanse of Iran. Prev Vet Med. 2013;109(1-two):162–vii.
-
Mirzaei M, Fooladi 1000. Coproscopy survey of gastrointestinal parasites in owned dogs of Kerman city, Iran. Vet Ital. 2013;49:309–13.
-
Gharekhani J. Study on gastrointestinal zoonotic parasites in pet dogs in Western Iran. Turkiye Parazitol Derg. 2014;38(three):172.
-
Arzamani Thou, Rouhani Southward, Mousazadeh-Mojarrad A, Sedeghi S, Rostami Thou, Raeghi Due south. Identification of zoonotic parasites isolated from stray dogs in Bojnurd County located in northward-east of Islamic republic of iran. NBM. 2016;four(iv):185–8.
-
Tavalla M, Kord E, Abdizadeh R, Asgarian F. Molecular study of cryptosporidium spp. in dogs from southwest of Iran. Jundishapur J Microbiol. 2017;10(four):e43412.
-
Mohaghegh M, Vafaei M, Ghomashlooyan M, Azami G, Falahati M, Azadi Y, et al. A wide diversity of zoonotic intestinal parasites in domestic and stray dogs in rural areas of Kermanshah Province, Islamic republic of iran. Trop Biomed. 2018;35(i):82–ninety.
-
Borji H, Razmi G, Movassaghi AR, Naghibi AG, Maleki M. Prevalence of cryptosporidium and Eimeria infections in dromedary (Camelus dromedarius) in abattoir of Mashhad, Iran. J Camel Pract Res. 2009;16(2):167–seventy.
-
Behzadi M, Razavi S, Yazdanpoor H, Mirzaei A, Tamadon A, Gandomani MJ. Epidemiology of cryptosporidium infection in ostriches (Struthio camelus) in Iran. Bulg J Vet Med. 2009;12(1):55–61.
-
Nazifi S, Behzadi Grand, Haddadi Due south, Jahromi AR, Mehrshad S, Tamadon A. Prevalence of cryptosporidium isolated from dromedary camels (Camelus dromedarius) in Qeshm Isle, southern Iran. Comp Clin Pathol. 2010;xix(3):311–4.
-
Sazmand A, Rasooli A, Nouri M, Hamidinejat H, Hekmatimoghaddam S. Prevalence of cryptosporidium spp. in camels and involved people in Yazd Province, Iran. Iran J Parasitol. 2012;7(1):eighty–4.
-
Yakhchali G, Moradi T. Prevalence of cryptosporidium-similar infection in ane-humped camels (Camelus dromedarius) of northwestern Islamic republic of iran. Parasite: SFP. 2012;xix(i):71.
-
Radfar MH, Gowhari MA, Khalili K. Comparison of capture ELISA and modified Ziehl-Neelsen for detection of Cryptosporidium parvum in feces of camel (Camelus dromedarius) in Iran. Sci Parasito. 2013;14(3):147–52.
-
Shahraki F, Rasekh M. Prevalence of Cryptosporidium parvum in the cities of Sistan region by ELISA and evaluation of risk factors of season, age and sex; 2016. https://ganj.irandoc.air-conditioning.ir. Accessed 29 Feb 2019.
-
Hamedi HM, Soleymani Grand. Parasites of the rodent and blood vessels of the caput and neck of the abbey. HMJ. 2004;vii(3):123–seven.
-
Shirazi Due south, Mirsamadi N, Shahbazi P. Evaluation of cryptosporidiosis in human, calf and mice around Tabriz. Vet J Islamic Azad Uni. 2010;half-dozen(iii):53–8.
-
Bahrami F, Sadraei J, Frozandeh M. Molecular characterization of cryptosporidium spp. in wild rats of Tehran, Iran using 18 s rRNA gene and PCR_RFLP method. Jundishapur J Microbiol. 2012;3(5):486.
-
Borji H, Khoshnegah J, Razmi G, Amini H, Shariatzadeh M. A survey on intestinal parasites of golden hamster (Mesocricetus auratus) in the northeast of Iran. J Parasit Dis. 2014;38(3):265–8.
-
Valipour NR. Prevalence of cryptosporidium in wild brown rat (Rattus norvegicus) population at Shoushtar, Iran. Int Electron J Med. 2016;five(1):18–22.
-
Mohebali M, Zarei Z, Khanaliha K, Kia EB, Motavalli-Haghi A, Davoodi J, et al. Natural intestinal protozoa in rodents (Rodentia: Gerbillinae, Murinae, Cricetinae) in northwestern Islamic republic of iran. Iran J Parasitol. 2017;12(iii):382.
-
Banani M, Dadras H, Moazeni Joula G, Houshmandrad P. Serologic incidence of cryptosporidial infection in broiler flocks in Shiraz, Islamic republic of iran. Curvation Razi Ins. 2000;51:95–102.
-
Mirzaei Yard, Mohammadi Five, Fotouhi A. Prevalence of intestinal cryptosporidium infection in Kerman pigeons. Sci Res Iranian Vet J. 2008;4(2):115–21.
-
Norolahi Fard F, Asl N, Rezaei Seghinsara H. Prevalence of intestinal cryptosporidiosis in pigeons in Mashhad. Iranian J Vet Clin Sci. 2011;4(one):21–eight.
-
Shemshadi B, Bahadori SR, Mozafari A. Study on cryptosporidiosis incidence in broilers in Garmsar region, Iran. Comp Clin Pathol. 2011;20(2):143–9.
-
Haghbin Northward, Mousavi Due south, Ranjbar B, Mohammadi Thousand, Hoseini S. Frequency of cryptosporidium infection in broiler breeding flock of Ghaemshahr. Vet Res (Ghamsar Branch). 2011;7(1):ane–five.
-
Radfar MH, Asl EN, Seghinsara Hour, Dehaghi MM, Fathi S. Biodiversity and prevalence of parasites of domestic pigeons (Columba livia domestica) in a selected semiarid zone of Due south Khorasan, Iran. Tropical Anim Heaith Pro. 2012;44(2):225–nine.
-
Hamidinejat H, Jalali MHR, Jafari RA, Nourmohammadi Yard. Molecular determination and genotyping of cryptosporidium spp. in fecal and respiratory samples of industrial poultry in Iran. Asian Pac J Trop Med. 2014;vii(seven):517–xx.
-
Hashemzadeh Farhang H, Bahavarnia SP Sr. Investigation of cryptosporidium parasitic infection in native poultry around Tabriz city. Vet Clin Pathol. 2015;8(3):588–95.
-
Hashemzadeh F, Shahbazi P, Jafari R. Survey of cryptosporidium parasite infection in poultry farms around Tabriz. Vet Clin Pathol. 2014;8(1):411–6.
-
Shemshadi B, Ranjbar-bahadori Southward, Delfan-abazari M. Prevalence and intensity of parasitic infection in domestic ducks (Anas platyrhynchas) in Gilan Province, northern Iran. Comp Clin Pathol. 2017;26(one):165–7.
-
Soltanialvar Masood GZ. Frequency of infection of native turkeys with Cryptosporidum parasites in Dezful metropolis. The first National Conference on mutual diseases betwixt humans and animals. JVH. 2017;3:1–7.
-
Larki Southward, Alborzi A, Chegini R, Amiri R. A preliminary survey on gastrointestinal parasites of domestic ducks in Ahvaz, Southwest Iran. Iran J Parasitol. 2018;xiii(1):137.
-
Naghibi A, Vahedi H. Prevalence of cryptosporidial infection in equus caballus and man in Mashhad, Iran. Arch Razi Inst. 2002;54:101–6.
-
Tavasouli 1000, Sodagar Southward, Soltan A. A survey on cryptosporidial infection in equus caballus in Urmia area, northwestern Iran. Islamic republic of iran J Vet Res. 2007;8(1):86–ninety.
-
Ghadrdan A, Hamidienjat H, Alizadehnia P. A survey on frequency of equine cryptosporidiosis in Ahvaz. Vet Clin Path Tabriz. 2013;half dozen(4):1723–7.
-
Jasim G, Marhoon A. Prevalence and molecular assay of cryptosporidium spp isolated from wild and domestic birds. Acta Parasitol. 2015;6(2):65–70.
-
Kuhn RC, Rock CM, Oshima KH. Occurrence of cryptosporidium and giardia in wild ducks forth the Rio Grande River valley in southern New Mexico. Appl Environ Microbiol. 2002;68(ane):161–5.
-
Bomfim T, Gomes R, Huber F, Couto One thousand. The importance of poultry in environmental dissemination of cryptosporidium spp. Open up Vet Sci J. 2013;7(ane):12–seven.
-
Traub RJ, Robertson ID, Irwin PJ, Mencke Due north, Thompson RA. Canine gastrointestinal parasitic zoonoses in India. Trends Parasitol. 2005;21(1):42–8.
-
Bahrami A, Doosti A, Nahravanian H, Noorian A, Asbchin S. Epidemiological survey of gastro-intestinal parasites in devious dogs and cats. Aust J Bones Appl Sci. 2011;5:1944–8.
-
Mirzaei Yard, Fooladi 1000. Prevalence of intestinal helminthes in owned dogs in Kerman metropolis, Iran. Asian Pac J Trop Med. 2012;5(nine):735–7.
-
Tavassoli Yard, Javadi S, Soltanalinejad F, Rosouli S, Etminanfar R. Gastrointestinal parasites of pet dogs in Urmia metropolis. Vet J Pajouhesh Sazandegi. 2010;87:18–24.
-
Simpson J, Burnie A, Miles R, Scott J, Lindsay D. Prevalence of giardia and cryptosporidium infection in dogs in Edinburgh. Vet Rec. 1988;123(17):445.
-
Batchelor D, Tzannes Southward, Graham P, Wastling J, Pinchbeck Thousand, German A. Detection of endoparasites with zoonotic potential in dogs with gastrointestinal illness in the UK. Transbound Emerg Dis. 2008;55(ii):99–104.
-
Dubná Southward, Langrová I, Nápravník J, Jankovská I, Vadlejch J, Pekár S, et al. The prevalence of abdominal parasites in dogs from Prague, rural areas, and shelters of the Czech Commonwealth. Vet Parasitol. 2007;145(i-2):120–eight.
-
Koompapong K, Mori H, Thammasonthijarern N, Prasertbun R, Pintong A-R, Popruk S, et al. Molecular identification of cryptosporidium spp. in seagulls, pigeons, dogs, and cats in Thailand. Parasite. 2014;21:52.
-
Huber F, Bomfim T, Gomes R. Comparison betwixt natural infection by cryptosporidium sp., giardia sp. in dogs in two living situations in the west zone of the municipality of Rio de Janeiro. Vet Parasitol. 2005;130(one-2):69–72.
-
Yoshiuchi R, Matsubayashi M, Kimata I, Furuya M, Tani H, Sasai 1000. Survey and molecular label of cryptosporidium and giardia spp. in owned companion animate being, dogs and cats, in Nippon. Vet Parasitol. 2010;174(three-4):313–half dozen.
-
Gil H, Cano L, de Lucio A, Bailo B, de Mingo MH, Cardona GA, et al. Detection and molecular diversity of giardia duodenalis and cryptosporidium spp. in sheltered dogs and cats in northern Espana. Infect Genet Evol. 2017;fifty:62–nine.
-
Olabanji GM, Maikai BV, Otolorin GR. Prevalence and gamble factors associated with faecal shedding of cryptosporidium oocysts in dogs in the Federal Capital Territory, Abuja, Nigeria. Vet Med Int. 2016;2016:1–seven.
-
Titilincu A, Mircean Five, Achelaritei D, Cozma 5. Prevalence of cryptosporidium spp. in asymptomatic dogs past ELISA and take a chance factors associated with infection. Lucrari Stiinłifice. 2010;43(1):vii–12.
-
Zhou L, Fayer R, Trout JM, Ryan UM, Schaefer FW, Xiao 50. Genotypes of cryptosporidium species infecting fur-begetting mammals differ from those of species infecting humans. Appl Environ Microbiol. 2004;70(12):7574–7.
-
Webster JP, Macdonald DW. Cryptosporidiosis reservoir in wild chocolate-brown rats (Rattus norvegicus) in the Great britain. Epidemiol Infect. 1995;115(1):207–9.
-
Paparini A, Jackson B, Ward S, Young S, Ryan UM. Multiple cryptosporidium genotypes detected in wild black rats (Rattus rattus) from northern Australia. Exp Parasitol. 2012;131(4):404–12.
-
Lv C, Zhang 50, Wang R, Jian F, Zhang S, Ning C, et al. Cryptosporidium spp. in wild, laboratory, and pet rodents in China: prevalence and molecular characterization. Appl Environ Microbiol. 2009;75(24):7692–9.
-
Kvác 1000, Hofmannová L, Bertolino S, Wauters L, Tosi K, Modrý D. Natural infection with ii genotypes of cryptosporidium in red squirrels (Sciurus vulgaris) in Italy. Folia Parasitol. 2008;55(two):95.
-
Ng-Hublin JS, Singleton GR, Ryan U. Molecular characterization of cryptosporidium spp. from wild rats and mice from rural communities in the Philippines. Infect Genet Evol. 2013;sixteen:five–12.
-
Feng Y, Alderisio KA, Yang Westward, Blancero LA, Kuhne WG, Nadareski CA, et al. Cryptosporidium genotypes in wildlife from a New York watershed. Appl Environ Microbiol. 2007;73(20):6475–83.
-
Majewska Air-conditioning, Werner A, Sulima P, Luty T. Prevalence of cryptosporidium in sheep and goats bred on 5 farms in west-central region of Poland. Vet Parasitol. 2000;89(4):269–75.
-
Yang R, Jacobson C, Gordon C, Ryan U. Prevalence and molecular characterisation of cryptosporidium and giardia species in pre-weaned sheep in Australia. Vet Parasitol. 2009;161(1-2):19–24.
-
Wang Y, Feng Y, Cui B, Jian F, Ning C, Wang R, et al. Cervine genotype is the major cryptosporidium genotype in sheep in China. Parasitol Res. 2010;106(two):341.
-
Koyama Y, Satoh M, Maekawa K, Hikosaka K, Nakai Y. Isolation of Cryptosporidium andersoni Kawatabi type in a slaughterhouse in the northern island of Nihon. Vet Parasitol. 2005;130(3-4):323–half dozen.
-
Nguyen ST, Nguyen DT, Le DQ, Le Hua LN, Van Nguyen T, Honma H, et al. Prevalence and first genetic identification of cryptosporidium spp. in cattle in Central Viet Nam. Vet Parasitol. 2007;150(4):357–61.
-
Sevinc F, Irmak K, MSevinc M. The prevalence of Cryptosporidium parvum infection in the diarrhoiec and not-diarrhoeic calves. Rev Med Vet. 2003;154(five):357–62.
-
Trotz-Williams L, Martin D, Gatei Due west, Cama V, Peregrine A, Martin Southward, et al. Genotype and subtype analyses of cryptosporidium isolates from dairy calves and humans in Ontario. Parasitol Res. 2006;99(4):346–52.
-
Fayer R, Santín M, Xiao L. Cryptosporidium bovis n. sp. (Apicomplexa: Cryptosporidiidae) in cattle (Bos taurus). J Parasitol. 2005;91:624–9.
Acknowledgements
Not applicative.
Writer information
Affiliations
Contributions
This study was done by RG, ZK, SK, FFP, HS, NB, FE, and MMH. RG and MMH participated in the pattern of the study. Data collection was washed by RG, FFP, and NB. Interpretation and manuscript preparation were conducted by MMH and RG. HS, RG, and MMH participated in the data editing. SK performed the statistical analysis. Fe and ZK performed the coordination and helped with the drafting of the manuscript. The authors read and canonical the final version of the manuscript.
Corresponding author
Ideals declarations
Ethics approval and consent to participate
Non applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open up Access This commodity is licensed nether a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(south) and the source, provide a link to the Artistic Commons licence, and indicate if changes were made. The images or other tertiary party material in this article are included in the commodity's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article'southward Creative Commons licence and your intended use is non permitted by statutory regulation or exceeds the permitted employ, you will need to obtain permission directly from the copyright holder. To view a re-create of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Reprints and Permissions
About this article
Cite this commodity
Haghi, M.M., Khorshidvand, Z., Khazaei, S. et al. Cryptosporidium animal species in Iran: a systematic review and meta-assay. Trop Med Health 48, 97 (2020). https://doi.org/ten.1186/s41182-020-00278-ix
-
Received:
-
Accustomed:
-
Published:
-
DOI : https://doi.org/10.1186/s41182-020-00278-9
Keywords
- Cryptosporidium
- Abdominal parasites
- Animal
- Systematic review
- Iran
Source: https://tropmedhealth.biomedcentral.com/articles/10.1186/s41182-020-00278-9
Postar um comentário for "Cryptosporidiosis in Human and Animal a Systematic Review and Meta-analysis"